E-mail:public_yc@163.com
Study on upconversion of triplet-triplet annihilation
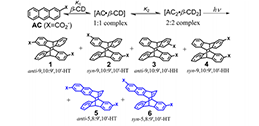
Up-conversion technology is a technology that converts low-energy light into high-energy light. Triplet-triplet annihilation up-conversion (TTA-UC) belongs to one of these types. Because of its advantages, such as low excitation light energy density and adjustable working wavelength, it is expected to be applied in the field of improving solar energy utilization efficiency. It is necessary to realize the practical application of up-conversion technology to obtain high up-conversion efficiency and to realize solid-phase up-conversion under air conditions. At present, optimizing the inherent photophysical properties of photosensitizers and receptors is the main method to improve the up-conversion efficiency of TTA.
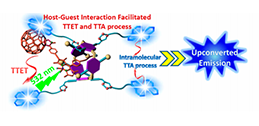
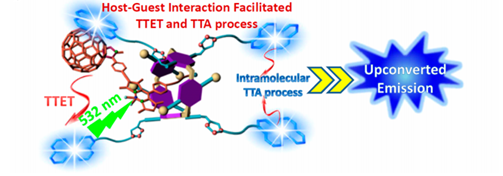
We established a supramolecular complexation based strategy for improving the efficiency of TTA-UC in the solution phase. The host−guest complexation facilitated the TTET and TTA processes between the sensitizers and emitters, and the TTA-UC intensity was significantly enhanced. This work opens a new window for improving TTA-UC efficiency without changing the inherent photophysical properties of the sensitizers and emitters.
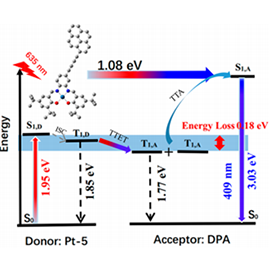
A series of Pt(II)–Schiff base complexes were synthesized as triplet sensitizers for the purpose of tuning the singlet and triplet energy levels so as to minimize energy loss during triplet–triplet annihilation (TTA) upconversion (UC). A deep-red to blue TTA-UC was achieved with an unprecedentedly large anti-Stokes shift of 1.08 eV. UC quantum yields of up to 21% (with a theoretical maximum efficiency of 50%) were observed in solution. The complexes also showed efficient UC emission in air-saturated hydrogels with a UC quantum yield up to 14.8%, which is much higher than the highest previously reported value. The low threshold excitation intensity provided by the present system offers promising potential for application in terrestrial solar energy conversion.
 |
 |
Pre:Study on supramolecular chiral compounds
Next:最后一页